|
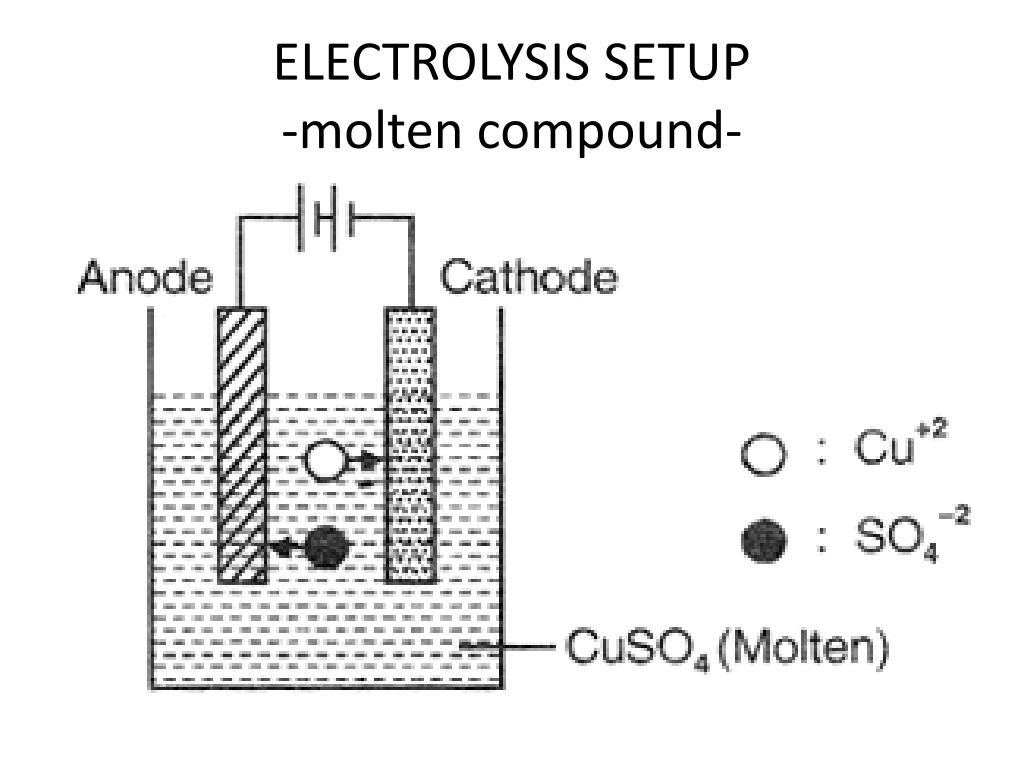
(a) Predict the products of the electrolysis and write the reactions occurring at the anode and cathode. In the electrolysis of CuCl2 solution using copper electrode, if 2.5 g of Cu is deposited at cathode, then at anode. The electrolysis was periodically stopped in order to adjust the compensated. A current of 19,0 A is passed through an electrolysis cell containing molten CaCl2 for 38.0 minutes. The cathode current density was varied in the range of 0.050.20 A cm 2 during the 24-hour potentiostatic electrolysis of the CaCl 2-CaF 2 melt in the electrolyser with screened catholyte containing the molybdenum cathode and the Al 2 O 3 powder (Fig.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
