|
When the substance behaves like an ideal gas, the ideal gas law describes the relationship between its pressure and volume. In the Unit on Thermochemistry, the relation between the amount of heat absorbed or related by a substance, q, and its accompanying temperature change, T, was introduced: q mcT (3.7.0.1) where m is the mass of the substance and c is its specific heat. Most phase changes occur at specific temperature-pressure combinations. We can examine aspects of the behavior of a substance by plotting a graph of pressure versus volume, called a PV diagram. Describe the relationship between heat (energy), bonding forces, and phase changes. 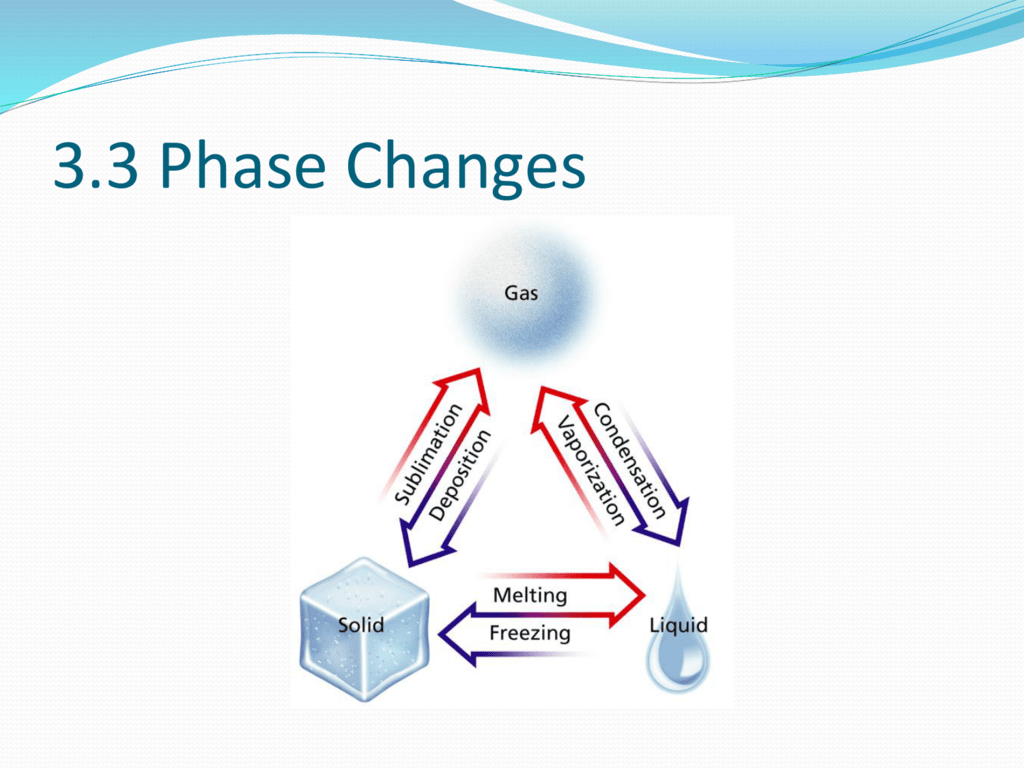 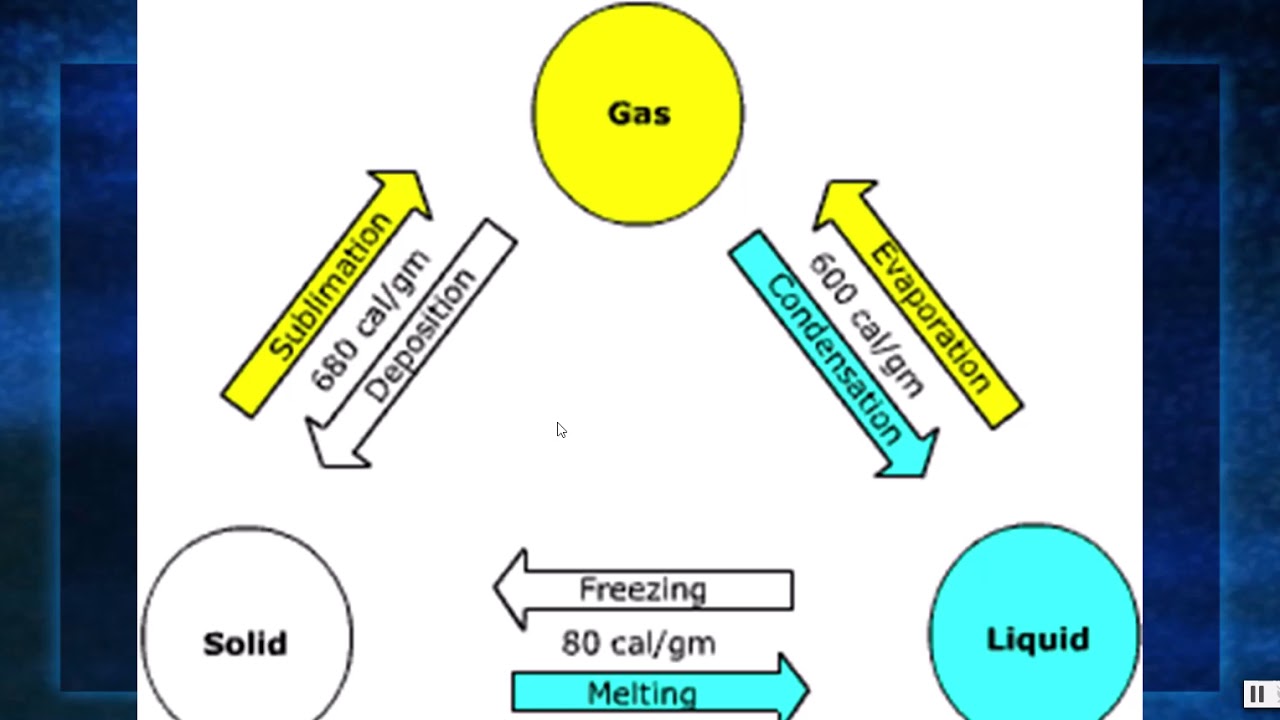
In dermatology, \(LN_2\) is used to freeze and painlessly remove warts and other growths from the skin. It is also used to reduce noise in electronic sensors and equipment, and to help cool down their current-carrying wires. \(LN_2\) is useful as a refrigerant and allows for the preservation of blood, sperm, and other biological materials. It boils at 77 K \((-196^oC)\) at atmospheric pressure. \(LN_2\) is made by liquefaction of atmospheric air (through compression and cooling). The four fundamental states of matter are solid, liquid, gas and plasma, but there others. Solid \(CO_2\) is called “dry ice.” Another example of a gas that can be in a liquid phase is liquid nitrogen \((LN_2)\). States of matter: Definition and phases of change. If the pressure is reduced, the temperature drops and the liquid carbon dioxide solidifies into a snow-like substance at the temperature \(-78^oC\). The amount of energy in molecules of matter determines the state of matter. Matter can exist in one of several different states, including a gas, liquid, or solid state. As long as you are at 100 C, you can change the phase by changing the pressure on the system. Imagine a vertical line through this diagram- for water, choose 100 degrees C. During evaporation, heat transfers into a substance, changing it from liquid to gas. For example, evaporation is an endothermic process. 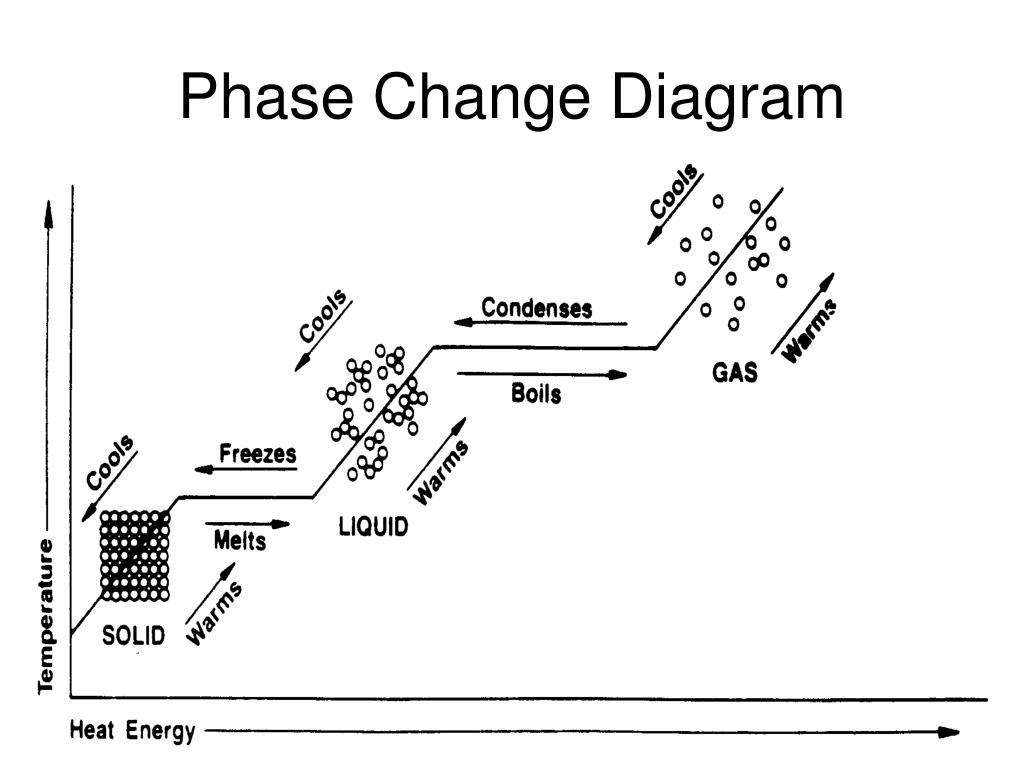
Carbon dioxide, for example, is a gas at room temperature and atmospheric pressure, but becomes a liquid under sufficiently high pressure. 7.3: Phase Changes 7.2: Heat and Temperature 7.4: Bond Energies and Chemical Reactions Anonymous LibreTexts Learning Objectives Determine the heat associated with a phase change. In the video here, Sal uses a horizontal line through the phase diagram. Phase changes can also be classified as exothermic or endothermic. High pressure may also cause a gas to change phase to a liquid. The volume decreases slightly once the substance is solid, but it never becomes zero. When the gas becomes a liquid, however, the volume actually decreases precipitously at the liquefaction point. The six most common phase changes are shown in Figure 11.5.1. The linear (straight line) part of the graph represents ideal gas behavior-volume and temperature are directly and positively related and the line extrapolates to zero volume at \(273.15^oC\), or absolute zero. These changes of state are often called phase changes. A change in the physical state of a substance. \): A sketch of volume versus temperature for a real gas at constant pressure. Matter exists in three physical states: Solid Liquid Gas When matter is converted from one physical state to another, a phase change occurs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
